 
Heavenly bodies, such as planets, are an option. germanium, americium, gallium, and so on. Elements also named for the places in which they were documented, e.g. The element einsteinium is a classic example. The origins of the element names generally fall into one of five major categories. The names of most of the elements have meaning and significance. Bromine and mercury are the only two elements in the periodic table that exist as liquids.ġ0. The alkali metals, alkaline earth metals, basic metals, transition metals, lanthanides, and actinides all are groups of metals. Most of the elements on the periodic table are metals. Elements within a group share several common properties and often have the same outer electron arrangement.ĩ. Columns of elements help to distinguish groups in the periodic table. Groups also show how an element can behave. For example, Sodium and Potassium are in the group I, so they both have one valence electron. Elements in each column have the same number of valence electrons. A group is also called a family of elements. 
Numerous synthetic radioisotopes of naturally occurring elements have also been produced in laboratories.Ĩ. Elements 95 to 118 have only been synthesized in laboratories or nuclear reactors. The first 94 elements, hydrogen through plutonium, all occur naturally, though some are found only in trace amounts and a few were discovered in nature only after having first been synthesized. The elements from hydrogen (H, atomic number 1) through oganesson (Uuo atomic number 118) were discovered or synthesized, completing seven full rows of the periodic table. Moreover, the radioactive element Uranium (U) is the heaviest element with an atomic weight of 238 in the periodic table. Technetium (Tc) was the first synthetic element in the periodic table of elements, which is the lightest element that has only radioactive isotopes. The element gallium (Ga) is a solid at room temperature but liquid above 29.7° Celsius. Therefore, he left blank spaces in his original periodic table so that he could properly line up the known elements. The element Gallium (Ga) was the first gap to be filled in 1875 by Mendeleev. Later, Ramsay proposed to Mendeleev that he added them to the table as a new column in 1890. For the most part, the order of the elements is the same between both tables, though there are exceptions.Ĥ.When William Ramsay and collaborators discovered the noble gases helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe) and radon (Rn) was not part of this periodic table of elements. The main difference between the modern periodic table and Mendeleev’s periodic table is that Mendeleev’s table arranged the elements in the order of increasing the atomic weight, while the modern table orders the elements by increasing the atomic number. This discovery strengthened his legacy as the father of the periodic table.ģ. In 1875, the discovery of gallium (Ga), scandium (Sc), and germanium (Ge) and their properties were matching with the predictions of Mendeleev. It was the first table that organized the elements according to periodic properties.Ģ. 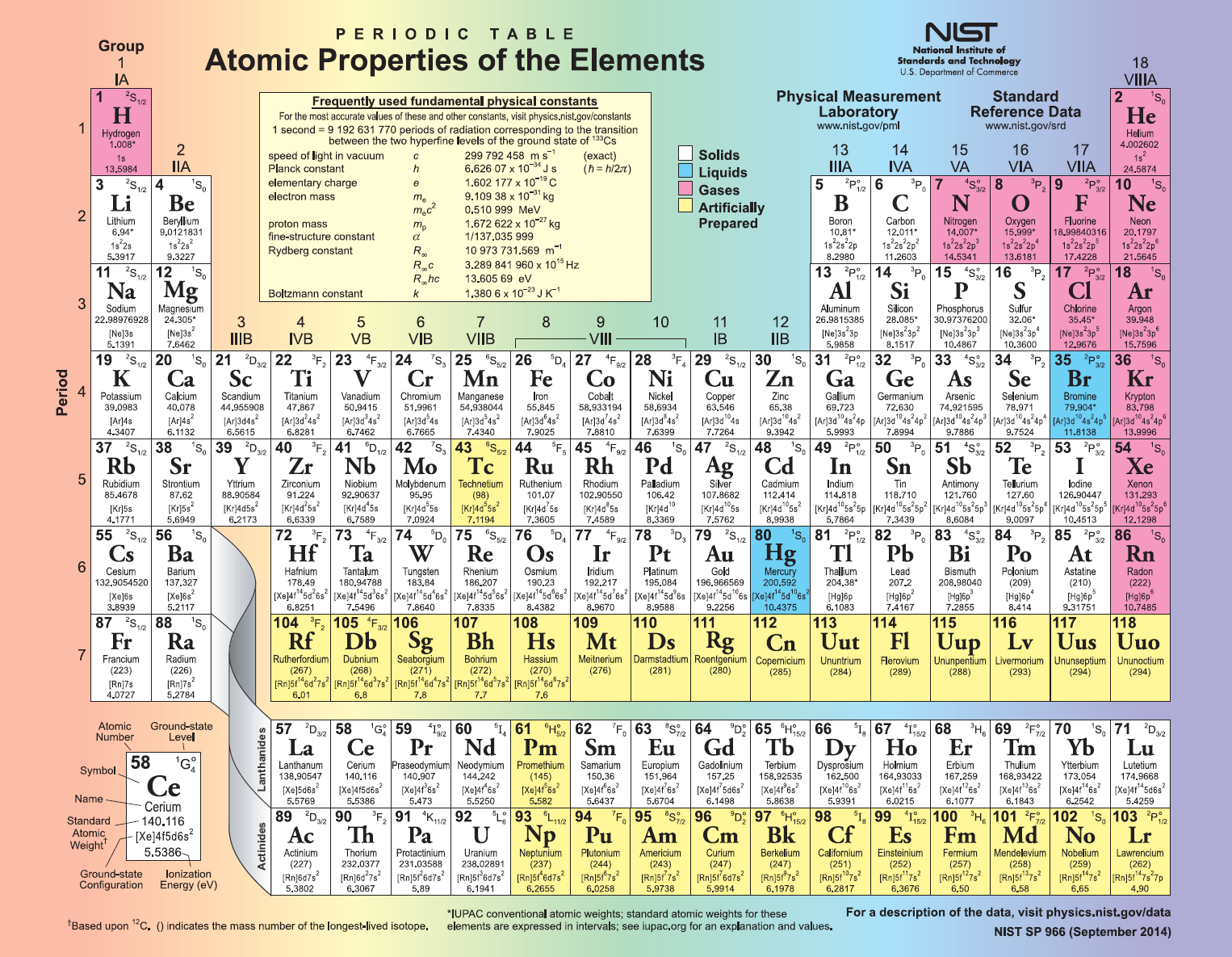
Dmitri Mendeleev is well known as the discoverer of the periodic table of elements, this table was the first to gain scientific credibility.Here are some fun facts regarding the periodic table of elements: Dmitri Mendeleev This periodic table of elements is one of the most powerful tools of chemistry and the other sciences. The rows of the table are called periods and the columns are called groups, which contain the elements with similar chemical behaviors. The structure of the table shows periodic trends. The periodic table of elements is a tabular display of the chemical elements that are arranged by atomic number, electron configuration, and persistent chemical properties. The periodic table of elements is one of the most important achievements in science, which is the essence not only of chemistry but also of physics and biology. Thus, the 150 th anniversary of Dmitry Mendeleev in 2019 was claimed as the “International Year of the Periodic Table of Chemical Elements” (IYPT2019). The modern periodic table of elements is a unique tool, enabling scientist to predict the appearance and properties of matter on the Earth and in the rest of the Universe. In 1869, Dmitri Mendeleev, the creator of the periodic table began collecting and sorting known properties of elements, as he was playing a card game, while traveling by train.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
